Particles from the air that have sufficient mass effect on the agar floor and viable organisms are permitted to mature out. A distant air intake is often used to reduce disturbance of your laminar movement industry.
Using wi-fi sensors and Online of Things (IoT) technology is enabling much more comprehensive and less intrusive environmental monitoring.
The pharmaceutical services that manufacture these merchandise are designed and created To maximise performance devoid of compromising product or service good quality or security. So where do you start if you are designing a non-sterile facility?
Staff Contamination: Gowning protocols make certain that human operators tend not to introduce contaminants into your cleanroom surroundings.
The applying of Federal Common 209E to clean rooms along with other controlled environments while in the pharmaceutical industry has actually been utilized by manufacturers of clean rooms to supply a specification for developing, commissioning, and protecting these facilities.
Really modest refrigerant charge applying R-448A. This ensures consumers Possess a long term proofed system that only needs a 6 lb. demand of refrigerant. This gets rid of the need for a bigger refrigerant charge and prospective leak risk.
Modular prepackaged Extra fat cooling heating device with variable speed evaporator admirers along with a variable velocity compressor leading to as much as 50% Strength discounts above regular procedure designs.*
The very first area where the Uncooked and packaging elements are subjected to the room atmosphere is from the incoming sampling room. Below containers should be opened to acquire samples of the Uncooked and packaging materials. The requirements for this region need to be similar to while check here in the production spot which is often ISO 8 Class 100,000. The transition position involving a warehouse and also the manufacturing area will be the dispensary or weigh room.
As we go to 2025, these conventional procedures will continue on to be significant, but their software might be more targeted and optimized.
Contamination threats—either by way of airborne microbes, personnel, or tools—can endanger solutions bringing about remembers or individual accidents. Thus, regimen fumigation is required to ruin residual microorganisms.
Yes, installation is available for the 4000 Collection Controlled Amount Chamber. FARRAR® also offers teaching and material freezing/thawing load screening for optimal velocity and uniformity throughout Procedure.
Temperature and Humidity: Environmental aspects like temperature and humidity are closely controlled to stop product degradation or microbial growth.
The key aim to get a design website staff is to reduce or remove the possibility of contamination inside of a cleanroom. One particular key to safety is acquiring airlocks between the process spot and the adjacent Area. This assists defend the products when becoming transferred from room to room.
Having said that, details available in the pharmaceutical industry deliver no scientific settlement over a connection in between the number of nonviable particulates and the focus of feasible microorganisms.
 Alicia Silverstone Then & Now!
Alicia Silverstone Then & Now!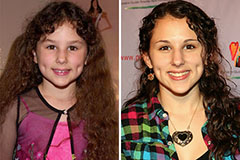 Hallie Eisenberg Then & Now!
Hallie Eisenberg Then & Now!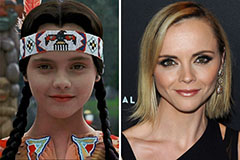 Christina Ricci Then & Now!
Christina Ricci Then & Now! Loni Anderson Then & Now!
Loni Anderson Then & Now!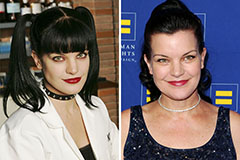 Pauley Perrette Then & Now!
Pauley Perrette Then & Now!